Portable Forced Oscillation Device for Pulmonary Function Testing
Cao, Jian, and Ashutosh Sabharwal. “Portable forced oscillation device for point-of-care pulmonary function testing.” Engineering in Medicine and Biology Society (EMBC), 2016 IEEE 38th Annual International Conference of the. IEEE, 2016.
TEAM MEMBERS:
Jian Cao, Graduate student, ECE
Gaurav Patel, Cognita Labs
Dr. Ashutosh Sabharwal, Professor, ECE
THE PROBLEM: Respiratory diseases have become a major health threat and affect nearly 10% of world’s population. To provide simple and robust lung function tests, a variety of methods have been developed. Among them, spirometry is the gold standard for measuring pulmonary functions. However, it is not suitable for several patient categories due to cooperation difficulty. As an alternative, the forced oscillation technique (FOT) measures the passive response of the respiratory system to external pressure oscillations. However, currently available FOT devices are bulky and high cost, and hence not often used.
OUR SOLUTION: In this study, we verify the feasibility of a smart and portable forced oscillation device based on a small subwoofer and ultrasonic sensor that is targeted at point-of-care pulmonary function testing.
To reduce the size of the device while ensuring clinical measurement accuracy, we build an ultrasonic sensor for high-resolution flow sensing without signal attenuation. We also optimize the signal processing algorithm for robust impedance estimation under low SNR. Furthermore, we develop an algorithm for automatic waveform adaptation and real-time error elimination. Finally, we evaluate the accuracy and repeatability of the device against both mechanical models and a small group of healthy human subjects.
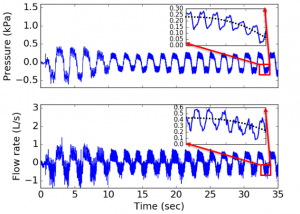
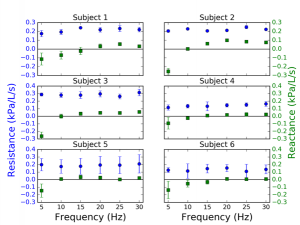
The results show that the error detection algorithm could successfully eliminate various artifacts including swallowing, glottis closure and abnormal breathing. With the adaptive oscillatory waveforms, the coherence function is above the clinical acceptance threshold 0.9 for all frequencies between 5Hz to 30Hz, and the relative measurement error of respiratory resistance is less than 10%. The device yields good repeatability and satisfies the clinical diagnostic requirements for point-of-care tests.