Modeling Aerosol Deposition
The Problem:
Metered Dose Inhalers (MDIs) are the most commonly used medication for treatment by the 510 million Asthma and COPD patients worldwide. The inhalers are essentially an aerosol spray that emits a metered amount of medication mixed with a propellant that can be inhaled into the lungs through the mouth. The efficiency of aerosol delivery to the lungs through this process is highly dependent on the patient’s technique of operating the MDI. However, the exact relationship between lung deposition of aerosol emitted from the MDI and the patient’s technique is unknown. Furthermore, the lung deposition is very expensive to measure and therefore, not practical during everyday use. Therefore, most patients continue to use MDIs unaware of the amount of medication actually being used by their body and hence, find it very difficult to keep their chronic pulmonary condition under control.
Modeling Aerosol Deposition

Our Solution:
We have designed an in-vitro experimental study to investigate the relationship between MDI use and lung deposition of aerosol emitted from the MDI. Our goal is to develop a predictive model based on our in-vitro experimental data to estimate lung deposition of aerosol for any MDI use technique.
In the first part of the project, in an IRB approved clinical study, we measured the actual MDI use from 23 asthma and COPD patients at the Pulmonary Function Testing lab at Ben Taub Hospital. The quantified MDI use data is the very first of its kind ever recorded, comprising of the complete breathing flow rate waveform, hand motion, and the MDI actuation timing called coordination, synchronized in time; coordination refers to the delay between the start of breathing and the time of pressing the MDI. The data was collected using the MDI monitoring device CapMedic. The results from the pilot study was presented at CHEST 2016; see publication [2] above. Based on the MDI use data collected from patients, we developed an MDI use model to represent the spectrum of MDI use techniques with varying trapezoidal flow waveforms and coordination.
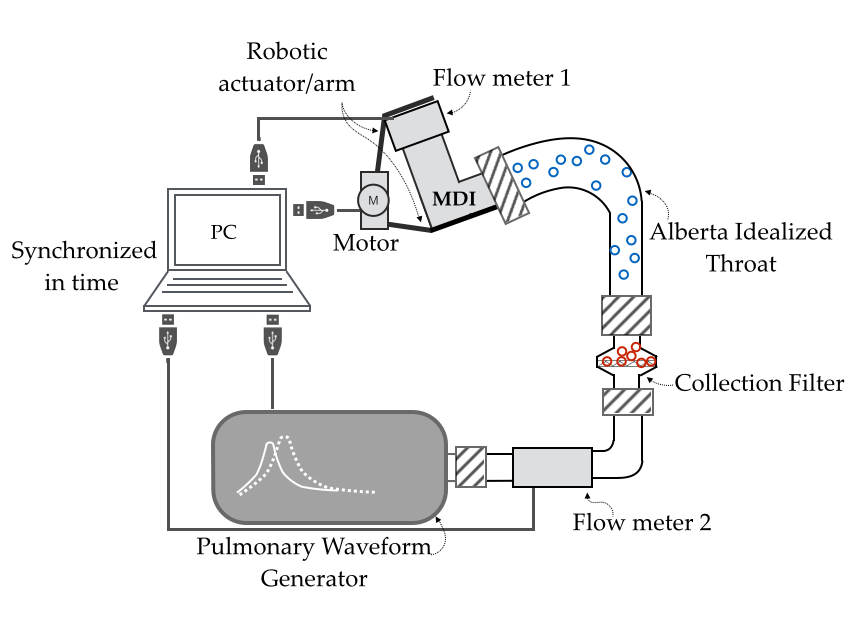
Second, we designed and developed a novel in-vitro experimental setup capable of emulating all parts of an MDI use, consisting of an idealized anatomical replica of the mouth-throat called Alberta Idealized Throat, collection filter (acting as the in-vitro lungs), a robotic MDI actuator for pressing the MDI, Pulmonary Waveform Generator (PWG) for generating breathing flow waveforms and flow meters for monitoring leakage. All the parts of the setup are synchronized to emulate the complete MDI use where the robotic arm actuates the MDI to release aerosol medication into the AIT connected to the collection filter, while the PWG generates breathing air flow. The experimental setup enabled for the first time, a study of two different MDI use parameters, coordination and non-uniform inspiration.
Our initial results, reported in [1], using the trapezoidal MDI use model, show that
- The lowest in-vitro lung deposition was 5.7%, for the case of no breathing during MDI use.
- A positive coordination that is, actuating the MDI after the start of inspiration, always resulted in higher in-vitro lung deposition compared with negative or zero coordination (actuating before or at the start of inspiration). Further, out of all parameters explored, variation in coordination affected lung deposition most significantly (by 23%).
- The in-vitro lung deposition for the case of high peak inspiratory flow rates (60-90L/min) resulted in higher in-vitro lung deposition as compared to low peak flow rates (30L/min).
Our results differ from the current GINA (Global Initiative for Asthma) inhaler use guidelines and suggest that we may need to investigate the current guidelines, which were designed for older inhalers.
Media Coverage (only select few listed):
- TV coverage on KPRC (local station)
- Wall Street Journal, story and video
- Futurity.org
- News Medical
- Technology.org
Team

Rajoshi Biswas
Graduate Student
ECE, Rice University

Nicola Hanania
Professor, Section of Pulmonary, Critical Care, and Sleep Medicine, Baylor College of Medicine

Dr. Ashutosh Sabharwal
Professor
ECE, Rice University
Paper
[1] Rajoshi Biswas, Nicola Hanania and Ashutosh Sabharwal, “Factors Determining In Vitro Lung Deposition of Albuterol Aerosol Delivered by Ventolin Metered Dose Inhaler,” Journal of Aerosol Medicin and Pulmonary Drug Delivery, 2017.
[2] Rajoshi Biswas, Gaurav Patel, Nicola Hanania and Ashutosh Sabharwal, “Measuring Competence in Metered Dose Inhaler Use Using CapMedic Electronic Inhaler Monitoring Tool” CHEST Journal 150.4_S (2016): 14A-14A.
